Experiment No.: 05
Experiment Name: Determination of Viscosity, Density, and Specific Gravity of Fuels.
Objectives:
- To determine the viscosity of different fuels using a viscometer.
- To determine the density and specificgravity of given fuels.
- To study the effect of fuel type on viscosity and density.
- To observe the effect of temperature on viscosity.
Introduction:
Viscosity, density, and specific gravity are important physical properties of fuels that significantly influence their flow behavior, atomization, combustion efficiency, and storage characteristics. In engineering applications, especially in internal combustion engines and fluid transport systems, accurate knowledge of these properties is essential. This experiment focuses on determining these properties for different fuels using standard laboratory instruments such as a rotational viscometer and a measuring cylinder.
Theory:
Viscosity is the property of a fluid that resists the relative motion between adjacent layers during flow. Due to internal friction, fluids with higher viscosity oppose motion more strongly. One important type of viscosity is kinematic viscosity, which represents the ability of a fluid to flow under gravity and is defined by
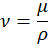
where is kinematic viscosity,
is dynamic viscosity, and
is density.
Density is defined as the mass per unit volume of a fluid and is expressed as
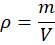
where is the mass and
is the volume of the fluid. Based on density, specific gravity is defined as the ratio of the density of a fuel to the density of water at the same temperature.
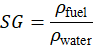
Temperature has a strong effect on viscosity. As temperature increases, liquid viscosity decreases due to reduced intermolecular attraction. Fuel type also influences viscosity, density, and specific gravity, where heavier fuels like engine oil show higher values compared to lighter fuels such as diesel, affecting fuel flow, lubrication, and combustion. In this experiment, viscosity is measured using a rotational viscometer in which a rotating spindle immersed in the fluid produces a resisting torque, indicated by the deflection of an analog dial meter and converted to viscosity using a calibrated scale.
Apparatus and Raw Materials:
- Viscometer
- Measuring Cylinder
- Weighing Balance
- Thermometer
- Water
- Fuels: Diesel, Engine Oil
Experimental Procedure:
- The viscometer was placed on a stable, horizontal surface and properly leveled.
- The fuel sample was poured into a clean container up to the specified level.
- A suitable spindle was selected and immersed vertically in the fuel without touching the container.
- The viscometer was operated at a constant rotational speed until the dial pointer became steady.
- The viscosity value was noted from the calibrated scale.
- 50 ml of the fuel was taken in a measuring cylinder.
- The mass of the measured fuel was determined using a weighing balance.
- Density was calculated from the ratio of mass to volume.
- The same procedure was repeated for all fuel samples.
Observation Table:
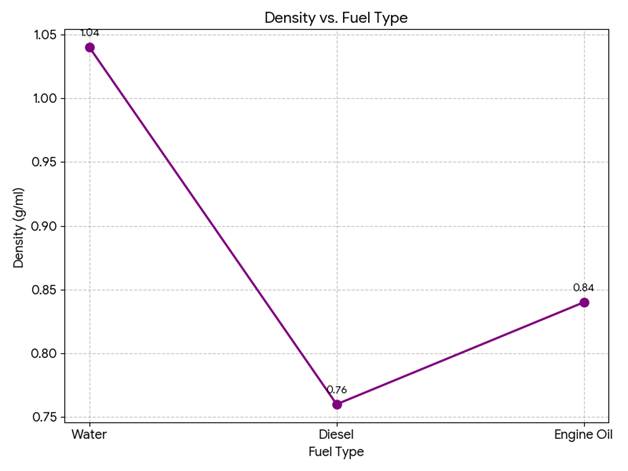
Table 1: Table for Viscosity, Density, and Specific Gravity of Fuels
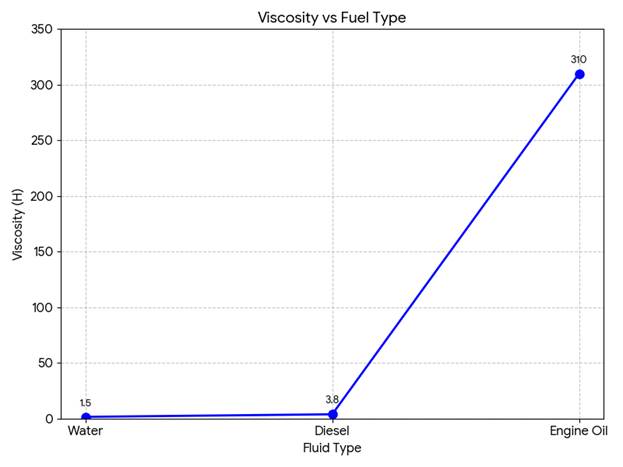
| Sl. No. | Fluid | Temperature (°C) | Viscosity (H) | Volume (ml) | Mass (g) | Density (g/ml) | Specific Gravity |
| 1 | Water | 18 | 1.5 | 50 | 52 | 1.04 | 1.04 |
| 2 | Diesel | 18 | 3.8 | 50 | 38 | 0.76 | 0.73 |
| 3 | Engine Oil | 18 | 310 | 50 | 42 | 0.84 | 0.81 |
Calculations:
For Water:
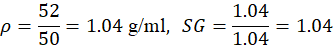
For Diesel:
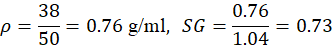
For Engine Oil:
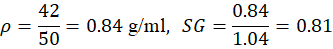
Graphs:
- Viscosity vs Fuel Type
- Density vs Fuel Type
(Graphs should be drawn manually on graph paper using experimental values.)
Results:
- The viscosity of engine oil was found to be the highest 310 H, followed by diesel and water.
- Water showed the highest density of 1.04 g/ml, while diesel had the lowest density of 0.76 g/ml.
- Specific gravity values confirmed that diesel and engine oil are lighter than water.
Discussion:
The experiment demonstrates that heavier fuels possess higher viscosity and density due to stronger intermolecular forces. Engine oil exhibited maximum resistance to flow, making it suitable for lubrication purposes. Diesel, being lighter, showed moderate viscosity. The controlled temperature ensures reliable measurements, as viscosity is highly temperature dependent. Minor experimental errors may arise due to heat loss and timing inaccuracies.
Conclusion:
The viscosity, density, and specific gravity of water, diesel, and engine oil were successfully determined using a viscometer and standard laboratory techniques. The experiment verified that viscosity and density depend strongly on fuel type and temperature. The obtained results closely align with standard reference values, confirming the accuracy of the experimental method.