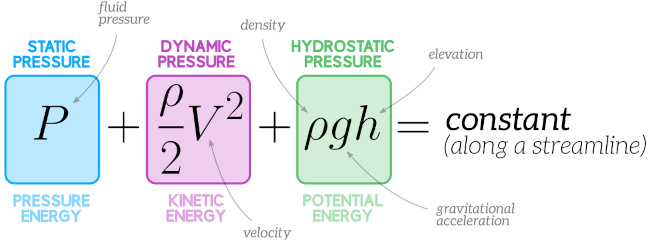
If the pressure of a substance is increased during a boiling process, will the temperature also increase or will it remain constant? Why?
Ans: If the pressure of a substance increases during a boiling process, the temperature also increases since the boiling (or saturation) temperature of a pure substance depends on pressure and increases with it. We are assuming that the liquid will continue to boil. If the pressure is increased fast enough, boiling may stop until the temperature has time to reach its new (higher) boiling temperature. A pressure cooker uses this principle.